- Accreditations
- Company
- Resources
- Chemical Analysis And Testing Services
- Mechanical Testing Services
- Metallurgical Testing and Metallography Services
- Fatigue Testing Services
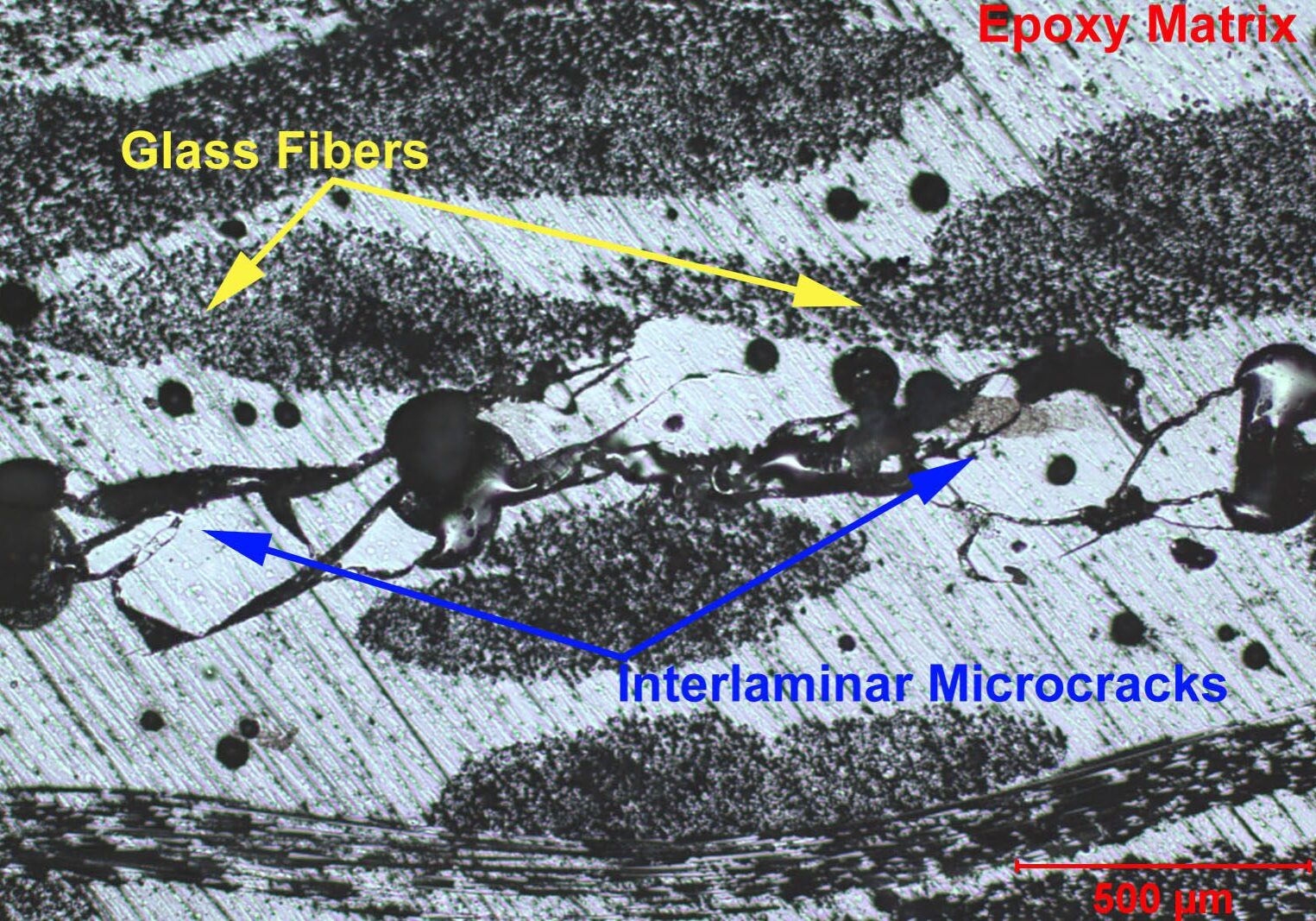
- Composite Testing Services
- Failure Analysis Services
- Coatings Testing and Evaluation Services
- Corrosion Testing and Salt Spray Services
- Engineering Support Services
- Blog/News
×
- Accreditations
- Company
- Resources
- Chemical Analysis And Testing Services
- Mechanical Testing Services
- Metallurgical Testing and Metallography Services
- Fatigue Testing Services
- Composite Testing Services
- Failure Analysis Services
- Coatings Testing and Evaluation Services
- Corrosion Testing and Salt Spray Services
- Engineering Support Services
- Blog/News